SCHEDULE
Dosing Information for SARCLISA
Recommended dose1
actual body weight administered as an IV infusion in combination with Kd or Pd
fixed infusion volume
Treatment is repeated until disease progression or unacceptable toxicity
IV=intravenous; Kd=carfilzomib and dexamethasone; Pd=pomalidomide and dexamethasone.
Infusion time decreases to 75 minutes by the third infusion1
Incremental escalation of the infusion rate should be considered only in the absence of IRRs.
IRR=infusion-related reaction.
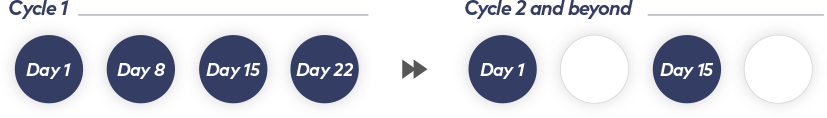
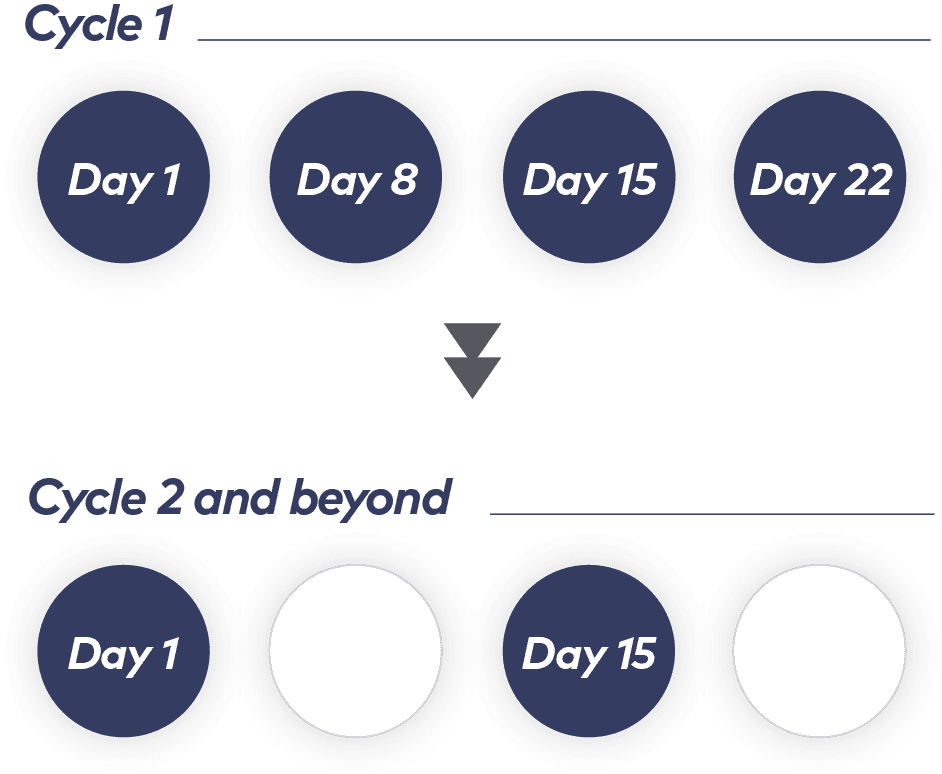
Weekly dosing transitions to every other week after the first cycle1
Treatment is administered in 28-day cycles
On days where both SARCLISA and carfilzomib are administered, administer dexamethasone first, followed by SARCLISA infusion, then followed by carfilzomib infusion.
For additional dosing instructions for combination agents administered with SARCLISA, refer to the study design pages for ICARIA-MM and IKEMA, and the respective manufacturer's Prescribing Information.
Premedication and antiviral prophylaxis1
Administer the following premedications prior to SARCLISA infusion to reduce the risk and severity of IRRs.
Dexamethasone
20 mg (IV on the days of SARCLISA and/or carfilzomib infusions, and orally on the other days) when administered in combination with SARCLISA and carfilzomib
40 mg orally or IV (or 20 mg orally or IV for patients ≥75 years of age) when administered in combination with SARCLISA and pomalidomide
Acetaminophen
650 mg to 1000 mg orally (or equivalent)
H2 antagonists
Institution-preferred agent
Diphenhydramine
25 mg to 50 mg orally or IV (or equivalent). The IV route is preferred for at least the first 4 infusions
The above recommended dose of dexamethasone (orally or IV) corresponds to the total dose to be administered only once before infusion as part of the premedication and of the backbone treatment, before SARCLISA and carfilzomib or SARCLISA and pomalidomide administration.
Administer the recommended premedication agents 15 to 60 minutes prior to starting a SARCLISA infusion. No post-infusion medications are required for SARCLISA.
Prophylaxis for herpes zoster reactivation
Initiate antiviral prophylaxis to prevent herpes zoster reactivation based on standard guidelines.
Infusion rates of SARCLISA administration1
Calculate the dose (mg) of required SARCLISA based on actual patient weight (measured prior to each cycle to have the administered dose adjusted accordingly). Note that more than one vial of SARCLISA may be necessary to obtain the required dose for the patient.
Incremental escalation of the infusion rate should be considered only in the absence of IRRs.
SARCLISA should be administered by a healthcare professional, with immediate access to emergency equipment and appropriate medical support to manage IRRs if they occur.
75-minute infusion time
starting after the second infusion in the absence of IRRs1
Administering SARCLISA
Prepare the solution for infusion using an aseptic technique1
- Administer the infusion solution by IV
infusion using an IV tubing infusion set (in PE, PVC with or without DEHP, PBD, or PU)
with a 0.22-micron in-line filter (PES, polysulfone, or nylon)
- The infusion solution should be administered for a period of time that will depend on the infusion rate (see the table above)
- Use prepared SARCLISA infusion solution within 48 hours when stored refrigerated at 36°F to 46°F (2°C to 8°C), followed by 8 hours (including the infusion time) at room temperature
- Do not administer SARCLISA infusion solution concomitantly in the same IV line with other agents
- On days where both SARCLISA and carfilzomib are administered, administer dexamethasone first, followed by SARCLISA infusion, then followed by carfilzomib infusion